EVENTS
HOSPITALAR 2024
Hospitalar 2024 (São Paulo, Brazil, 21-24 May) is just around the corner.
We are thrilled to announce that Cedic will be showcasing. Look for us at booth F-193!
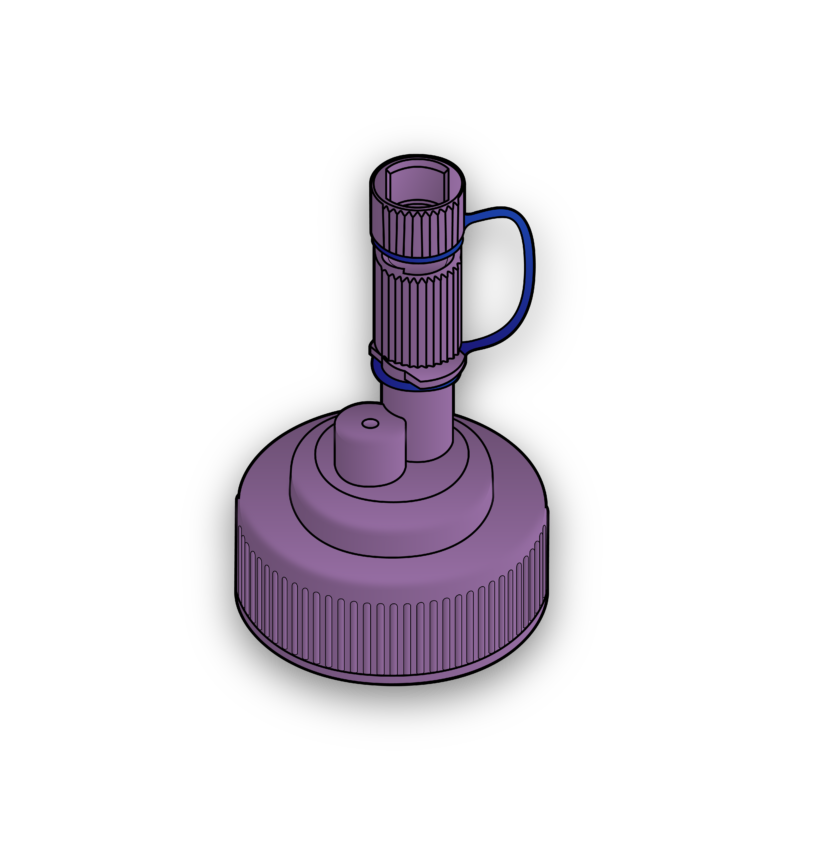
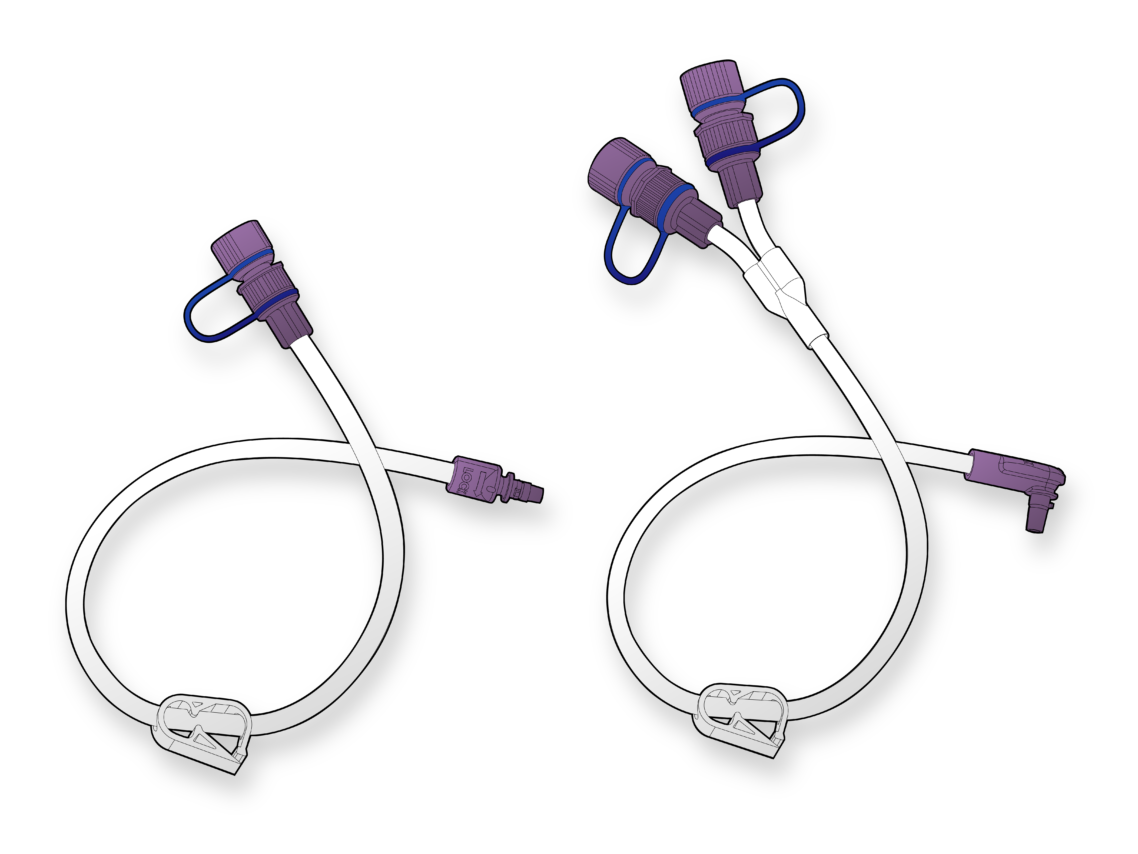
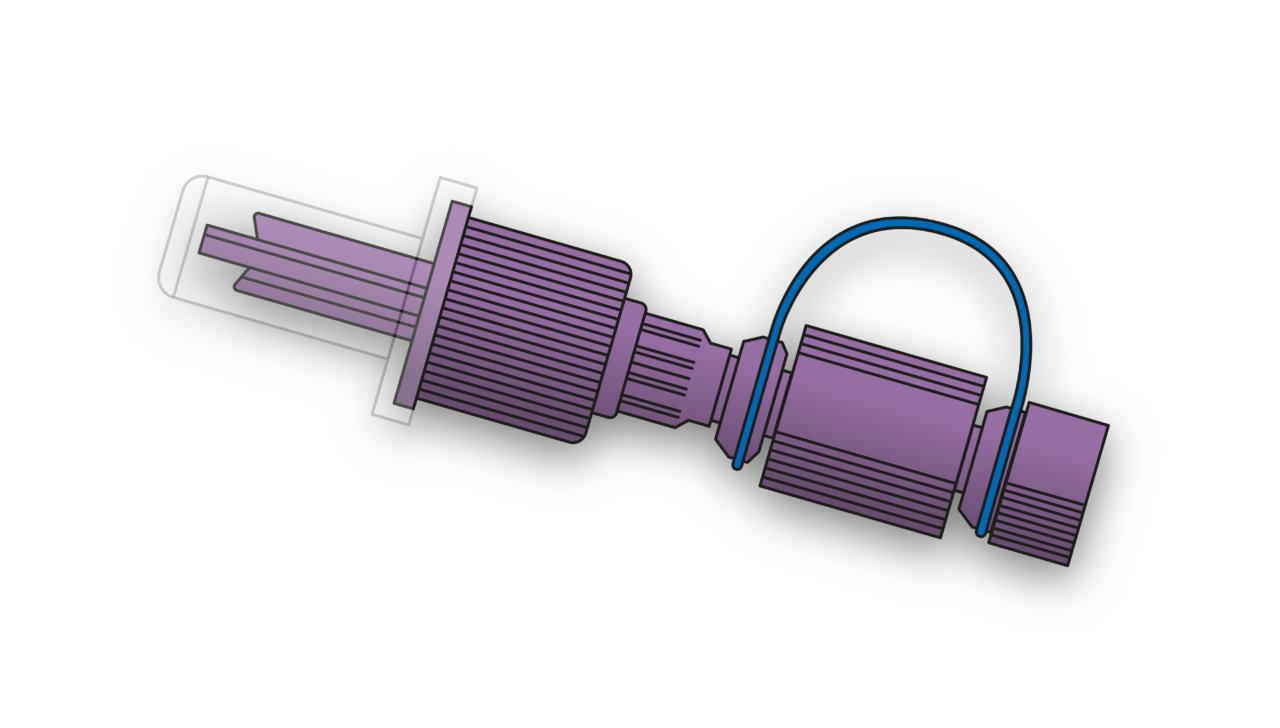
Join us to explore our solutions in devices and components for enteral nutrition and fluid delivery.
It’s a fantastic opportunity to connect, exchange insights, and discover how we’re shaping the future of healthcare together.
Looking forward to seeing you there!